-
Publish Your Research/Review Articles in our High Quality Journal for just USD $99*+Taxes( *T&C Apply)
Offer Ends On
Eren Kutay Zeyrek*, Talha Sonceley, Nedim Ozer and Hakki Tanyeri
Corresponding Author: Eren Kutay Zeyrek, Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Istanbul Kent University, Istanbul, Turkey.
Received: February 20, 2026 ; Revised: February 24, 2026 ; Accepted: February 30, 2026 ; Available Online: March 06, 2026
Citation: Zeyrek EK, Sonceley T, Ozer N & Tanyeri H. (2026) Medication-Related Osteonecrosis of the Jaw Following Dental Implant Surgery in a Patient Receiving Denosumab: A Case Report. J Oral Health Dent Res, 5(2): 1-8.
Copyrights: ©2026 Zeyrek EK, Sonceley T, Ozer N & Tanyeri H. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Views & Citations
Likes & Shares
Denosumab is a human monoclonal IgG2 antibody that induces changes in bone by inhibiting the differentiation and function of osteoclast precursor cells. As an anti-resorptive medication, it can potentially lead to medication-related osteonecrosis of the jaw (MRONJ). It is used to reduce bone-related events associated with metastases in cancers such as prostate cancer. Side effects of this drug include hypocalcemia and osteonecrosis of the jaw. It is important for clinicians to be well-versed in all aspects of MRONJ in order to make accurate diagnoses, provide appropriate care, and manage and prevent complications. This case describes the diagnosis and treatment approach of osteonecrosis that developed after dental implant surgery in a 79-year-old male patient who was treated with subcutaneous denosumab (an anti-resorptive agent) for prostatic adenocarcinoma. The importance of these cases in the field of dentistry is also discussed.
Keywords: Medication-related osteonecrosis of the jaw, MRONJ, Denosumab, Dental implant, Sequestrectomy
INTRODUCTION
Medication-related osteonecrosis of the jaw (MRONJ) is a severe condition characterized by exposed bone areas that persist for more than eight weeks and progressive bone destruction in the jaws, resulting from the use of antiangiogenic and antiresorptive medications [1,2]. Examples of these drugs include zoledronate, ibandronate, alendronate, and risedronate from the bisphosphonate group; denosumab from the RANKL inhibitor group; sunitinib from the tyrosine kinase inhibitor group; and bevacizumab from the monoclonal antibody group [2].
Clinically, patients may have osteonecrosis, intraoral or extraoral fistulas, purulent drainage, halitosis, pain, swelling, and pathologic fractures [3]. The prevalence of MRONJ was 0.043% in patients treated with oral bisphosphonates, 1.03% in those on intravenous bisphosphonates, and 3.64% in those on high-dose denosumab [4]. Denosumab (DMB) is an antiresorptive agent that acts as a receptor activator of nuclear factor kappa-B ligand (RANK-L), a fully humanized antibody that inhibits osteoclast function and associated bone resorption. When administered subcutaneously every six months, Denosumab (Prolia) significantly reduces the risk of vertebral, non-vertebral, and hip fractures in patients with osteoporosis. [5-8].
In addition, Denosumab (Xgeva) is effective in reducing skeletal-related events (SREs) due to metastatic bone disease from solid tumors and is administered monthly. Denosumab was more effective than zoledronic acid in preventing skeletal-related events and represents a potential new treatment option for men with bone metastases from castration-resistant prostate cancer [9].
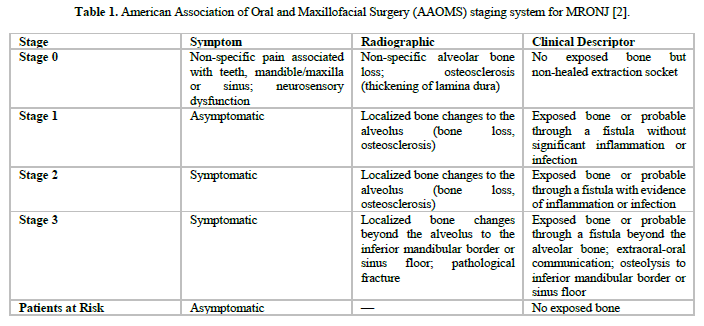
The AAOMS has developed a four-tier classification system for Medication-Related Osteonecrosis of the Jaws (MRONJ). This classification is used to determine disease severity and guide appropriate treatment strategies (Table 1). The American Association of Oral and Maxillofacial Surgery (AAOMS) recommends conservative treatment for MRONJ for stages 0 and 1, superficial debridement for stage 2 and surgical debridement and resections for stage 3 [2].
CASE REPORT
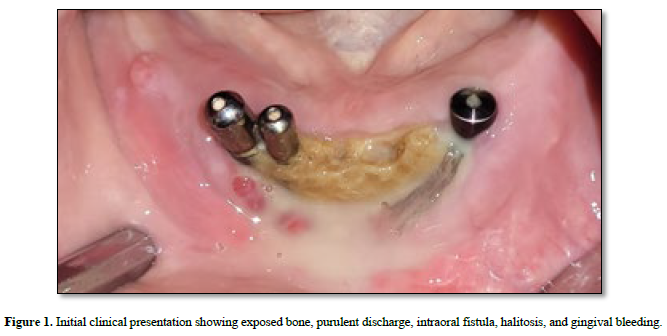
A 79‑year‑old male presented to our clinic with exposed bone in the mandibular symphysis, purulent discharge, pain, halitosis, and gingival bleeding (Figure 1). His medical history was notable for prostate adenocarcinoma treated with X‑Geva® (denosumab), Eligard® (leuprolide acetate), and Taxotere® (docetaxel). Dental implants had been placed in the anterior mandible during the period of antiresorptive therapy.
Following detailed anamnesis and clinical examination, for a more comprehensive evaluation, cone‑beam computed tomography (CBCT) and bone scintigraphy were obtained.
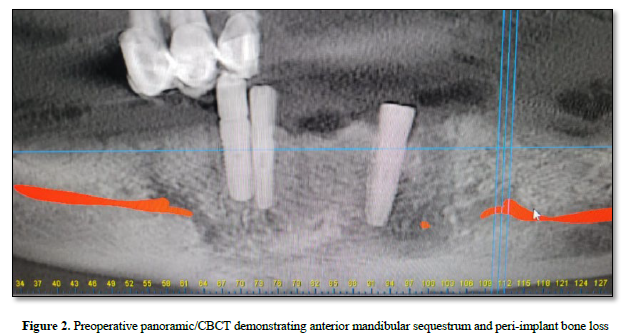
The scintigraphy report described increased osteoblastic activity in the anterior mandible, bilaterally extending from the midline, corresponding to the area of prior dental intervention, consistent with osteonecrosis. And CBCT demonstrated a sequestrum in the anterior mandible with peri‑implant bone loss (Figure 2).
The overall picture supported the diagnosis of medication-related osteonecrosis of the jaw (MRONJ).
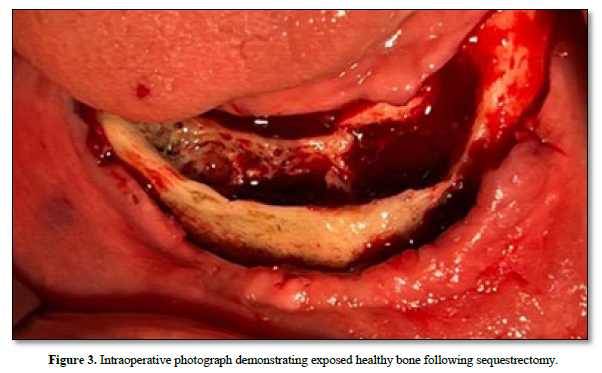
Following these conditions, sequestrectomy was planned as the appropriate treatment. To improve surgical safety, the patient was followed for four weeks with daily isotonic irrigation and local dressing. Once relevant consultations were obtained and the clinical condition was considered acceptable, the patient was scheduled for surgery.
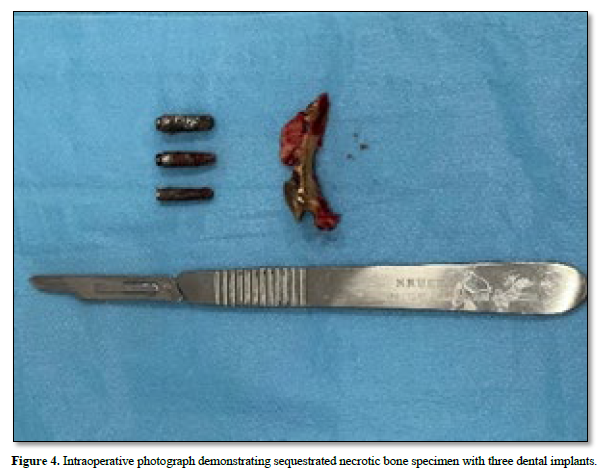
Under general anesthesia with nasal intubation, the procedure was started to perform an appropriate and well-planned sequestrectomy. The necrotic bone, which separated easily from the surrounding healthy bone, was carefully removed together with three implants located in the lesion (Figures 3,4). Generous irrigation with isotonic saline was applied to the surgical field to eliminate debris and reduce the risk of infection. After debridement and confirmation of physiological intraosseous bleeding indicating healthy bone (Figure 3), passive and tension-free primary closure was achieved.
The removed specimen (Figure 4) was sent entirely for histopathological examination, which confirmed necrotic bone without evidence of malignancy. According to the pathological report, the specimen showed fragmented necrotic bone together with granulation tissue, consistent with osteonecrosis with associated acute inflammatory changes.
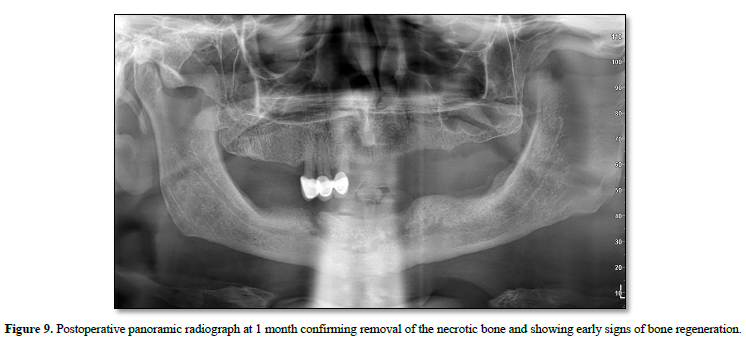
Postoperatively, the patient received systemic antibiotics, nonsteroidal anti-inflammatory drugs, a short course of corticosteroids, and chlorhexidine mouth rinses. Early postoperative follow-up demonstrated favorable soft tissue and bone healing with satisfactory clinical recovery. At the 1-month follow-up, the panoramic radiograph confirmed complete removal of the necrotic segment and showed early signs of bone regeneration (Figure 9).
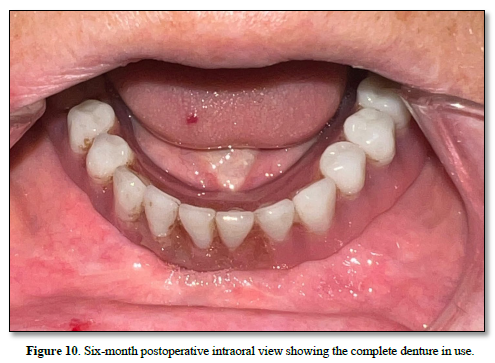
At six months, functional rehabilitation was achieved with a complete denture, which the patient reported using comfortably (Figure 10).
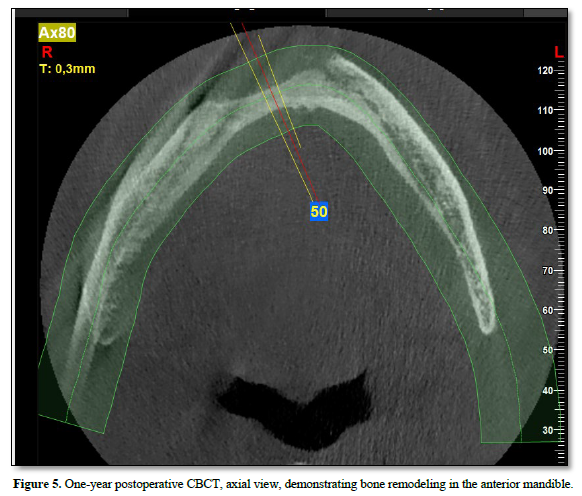
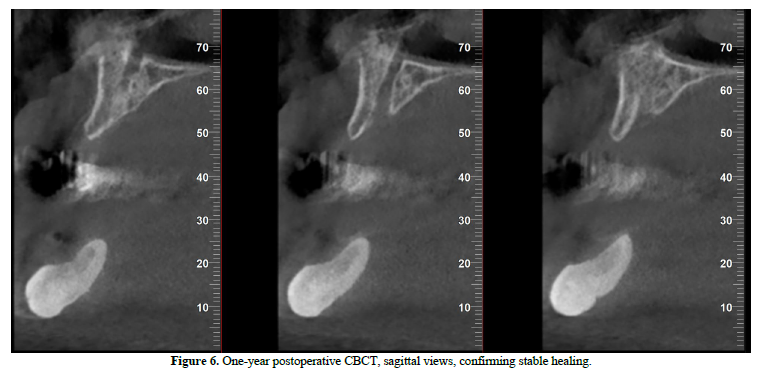
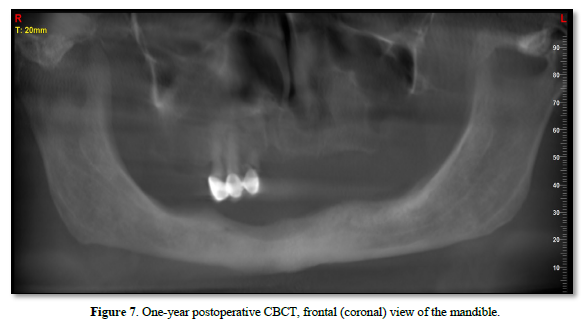
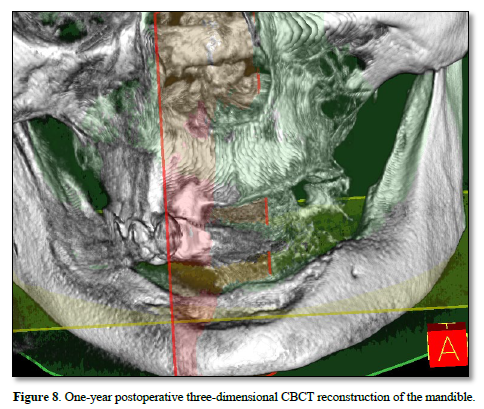
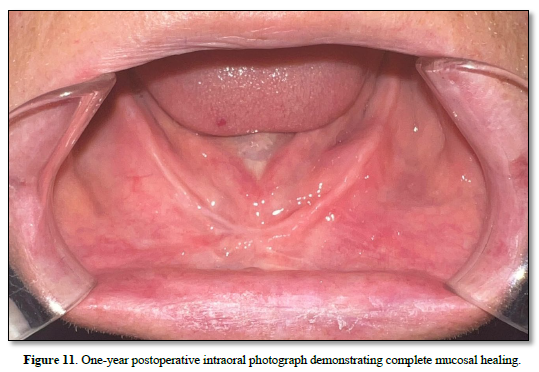
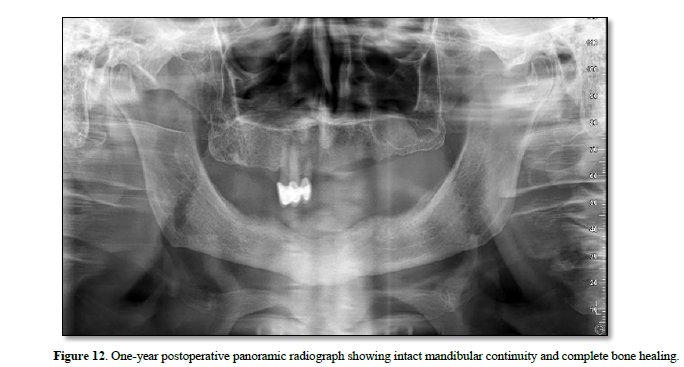
At the one‑year follow‑up, intraoral examination showed complete mucosal healing (Figure 11). CBCT acquired at the same visit—axial, sagittal, and frontal views plus three‑dimensional reconstruction—demonstrated stable bone remodeling of the anterior mandible (Figures 5–8). A one-year postoperative panoramic radiograph also confirmed preserved mandibular continuity, absence of recurrent pathology, and satisfactory bone healing (Figure 12).
The patient has remained asymptomatic for over 12 months with no clinical or radiologic evidence of recurrence.
DISCUSSION
Medication-related osteonecrosis of the jaw (MRONJ) is a serious complication associated with antiresorptive agents, including bisphosphonates and denosumab. Although the pathophysiology remains incompletely understood, inhibition of osteoclast-mediated bone remodeling and impairment of angiogenesis are considered pivotal mechanisms [10]. In our case, the patient developed stage II MRONJ in the anterior mandible following dental implant surgery during high-dose denosumab (Xgeva®) therapy for metastatic prostate adenocarcinoma. This aligns with previous reports that invasive dentoalveolar procedures, particularly extractions and implant placement, represent the most frequent precipitating events for MRONJ in denosumab-treated individuals [10,12].
Compared to bisphosphonates, denosumab exhibits a different pharmacokinetic profile, lacking bone binding and showing a reversible effect within approximately six months after discontinuation [13]. Despite this, its antiresorptive potency may lead to earlier onset of MRONJ, even after a small number of doses [12]. The present case underscores the importance of preoperative risk assessment and interdisciplinary consultation before surgical interventions in patients receiving denosumab. Notably, studies have reported a higher incidence of MRONJ in oncology patients receiving high-dose regimens (120 mg monthly) than in osteoporotic patients on lower doses (60 mg biannually) [10].
Management strategies for MRONJ vary depending on disease stage, patient comorbidities, and functional needs. While conservative measures such as chlorhexidine mouth rinses and systemic antibiotics are often first-line for early stages, accumulating evidence supports surgical intervention for favorable outcomes, even in advanced disease [10,12]. In our case, surgical debridement with implant removal and primary closure led to complete mucosal healing and symptom resolution, in accordance with recent prospective reports describing improved quality of life and high rates of remission after surgery [10].
The use of serum C-terminal telopeptide (CTX) levels as a predictive marker for MRONJ risk remains controversial. Some authors suggest that values <100 pg/mL may indicate high risk, advocating for the postponement of elective invasive dental procedures until levels recover [11]. In our patient, the CTX threshold (>100 pg/mL) guided surgical timing, potentially contributing to the uneventful postoperative course [11]. Nonetheless, robust clinical trials are lacking, and individualized patient assessment remains essential.
In conclusion, this case reinforces the necessity for heightened vigilance when planning oral surgical procedures in patients undergoing denosumab therapy, especially at oncologic dosages. Thorough preoperative evaluation, appropriate timing of intervention, and a multidisciplinary approach are crucial to reducing MRONJ risk and optimizing patient outcomes. Further longitudinal studies are warranted to refine prevention protocols, clarify the role of CTX monitoring, and compare the long-term efficacy of conservative versus surgical management in denosumab-associated MRONJ.
CONCLUSION
In patients receiving denosumab therapy, thorough clinical and radiographic evaluation, accurate timing, and appropriate surgical planning are critical to minimizing the risk of MRONJ development. Particularly in those undergoing high-dose oncologic regimens, meticulous assessment of risk factors and a multidisciplinary team approach are essential to prevent potential complications before surgical interventions. During the treatment process, patient education, optimization of oral hygiene, and regular follow-up visits not only enhance the effectiveness of preventive strategies but also enable early-stage intervention when necessary [1,2].
Moreover, comprehensive prospective clinical studies are needed to compare the long-term outcomes of surgical and conservative management approaches in MRONJ, to clarify the role of biochemical markers such as CTX in clinical decision-making, and to develop specific protocols tailored to identified risk groups. These future investigations will contribute to refining prevention strategies, improving treatment outcomes, and ultimately enhancing the quality of life for patients at risk of or affected by MRONJ [3,4].
No Files Found
Share Your Publication :